
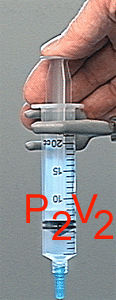
P1V1 = P2V2
Boyle's law describes the relationship between pressure and volume for a gas that undergoes compression or expansion at constant pressure.
 |
P1V1 = P2V2 Boyle's law describes the relationship between pressure and volume for a gas that undergoes compression or expansion at constant pressure.
|
 |



