Biomolecules:
Carbohydrates
| Home |
| Table of Contents |
| Biomolecules Gateway Page |
| Jmol Tutorial |
In this module: |
| Introduction |
| Definition |
| Linear and Cyclic Forms |
| Anomers |
| Disaccharides |
| Polysaccharides |
| Variations |
| Other Functions |
Sources of Carbohydrates' Variation
Besides compounds with the molecular formula Cx(H2O)y, other molecules derived from such compounds are also considered carbohydrates. The most common modifications are to add sulfate, N-acetyl, or carboxyl groups.
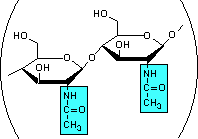
Think for a moment about the different variations there can be when carbohydrates are linked together to form polymers:
- Many different monosaccharides (glucose, galactose, and fructose, for example) can serve as the basic building blocks of the polysaccharide.
- These monosaccharides can be modified by the attachment of groups (acetyl, sulfate, and carboxyl, for example) at various carbon positions.
- The monosaccharides can be linked through several of their carbon positions (C1-C4, C1-C6, and C4-C6, being the most common).
- Finally, each glycosidic bond can be two different anomers, either α or β.
Thus, carbohydrates are information-rich molecules, and serve many different roles in the body, as the next page will show. Scientists are only beginning to unravel the ways carbohydrates store and use information in the body.
Variations