Biomolecules:
Lipids
Calorimetry
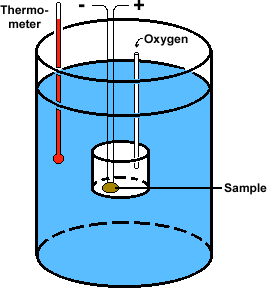 An important role of lipids is in energy storage. Scientists measure the energy stored in materials by a process called calorimetry. The instrument diagrammed at left is called a bomb calorimeter. The sample is placed in a small sealed can with electrical leads and an inlet for oxygen. This can is placed in a larger reservoir of water. Then the leads are used to ignite the sample. The energy released by the burning of the sample heats the water in the reservoir, and the temperature change is measured by the thermometer. Using the heat capacity of water, the energy released by burning the sample can be calculated.
An important role of lipids is in energy storage. Scientists measure the energy stored in materials by a process called calorimetry. The instrument diagrammed at left is called a bomb calorimeter. The sample is placed in a small sealed can with electrical leads and an inlet for oxygen. This can is placed in a larger reservoir of water. Then the leads are used to ignite the sample. The energy released by the burning of the sample heats the water in the reservoir, and the temperature change is measured by the thermometer. Using the heat capacity of water, the energy released by burning the sample can be calculated.
![]() How can you use heat capacity
How can you use heat capacity
to determine heat gain or loss?
![]() The bomb calorimeters below have 5.00 x 10-1 kg water reservoirs and 1.00 g of a sample, either vegetable shortening or table sugar. Click on the calorimeters to burn the samples and measure the temperature changes (you can click on them again to reset them to their initial conditions). Assuming that the calorimeters have been calibrated properly, calculate the heat (in kJ) released by the samples, enter your answers in the spaces below, and click on the Check button to check your answers.
The bomb calorimeters below have 5.00 x 10-1 kg water reservoirs and 1.00 g of a sample, either vegetable shortening or table sugar. Click on the calorimeters to burn the samples and measure the temperature changes (you can click on them again to reset them to their initial conditions). Assuming that the calorimeters have been calibrated properly, calculate the heat (in kJ) released by the samples, enter your answers in the spaces below, and click on the Check button to check your answers.
Calorimetry