|
GOALS:
After completing this module, you should be able to:
- Tell what enzymes can and cannot do
- Describe how enzymes accelerate specific chemical reactions
- Describe the difference between competitive and noncompetitive inhibition
AIDS and Protease Inhibitors
One of the most promising possibilities for a cure to the AIDS epidemic is a class of drugs called protease inhibitors. The goal of this mix of drugs is the inhibition of a specific enzyme produced by the AIDS virus, the HIV-1 protease. After completing this module, you should be able to understand how these drugs accomplish their goal.
Purpose of Enzymes
Literally millions of chemical reactions occur within each of our cells every minute. In the test tube, these reactions often proceed at an extremely slow rate. For example, the dissociation of carbonic acid into CO2 and H2O that takes place in the lungs:
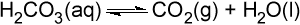
only proceeds at a rate of about 10-7 mol/L/s. CO2 is produced in the body at a much higher rate than this, however, and so some way to accelerate the reaction is needed.
As you have learned, raising the temperature is one way to accelerate reactions. The temperature needed to accelerate some reactions (hundreds of degrees Celsius) to the required speed is too high to be compatible with life, however.
The answer is to catalyze these reactions. Biological catalysts are known as enzymes
 , and the vast majority of enzymes are proteins. For example, the enzyme carbonic anhydrase accelerates the above reaction at room temperature by more than a million times the rate of the uncatalyzed reaction. In this module you will learn something about how enzymes work.
, and the vast majority of enzymes are proteins. For example, the enzyme carbonic anhydrase accelerates the above reaction at room temperature by more than a million times the rate of the uncatalyzed reaction. In this module you will learn something about how enzymes work.
|